Bamberger rearrangement
Appearance move to sidebar hideThe Bamberger rearrangement is the chemical reaction of phenylhydroxylamines with strong aqueous acid, which will rearrange to give 4-aminophenols. It is named for the German chemist Eugen Bamberger (1857–1932).
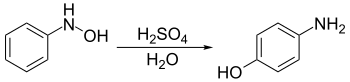 The Bamberger rearrangement
The Bamberger rearrangement
The starting phenylhydroxylamines are typically synthesized by the transfer hydrogenation of nitrobenzenes using rhodium or zinc catalysts.
One application is in the synthesis of fenhexamid.
Reaction mechanism
The mechanism of the Bamberger rearrangement proceeds from the monoprotonation of N-phenylhydroxylamine 1. N-protonation 2 is favored, but unproductive. O-protonation 3 can form the nitrenium ion 4, which can react with nucleophiles (H2O) to form the desired 4-aminophenol 5.
 The mechanism of the Bamberger rearrangement
The mechanism of the Bamberger rearrangement
See also
- Friedel–Crafts alkylation-like reactions:
- Bamberger triazine synthesis — same inventor
References
- ^ Harman, R. E. (1955). "Chloro-p-benzoquinone" (PDF). Organic Syntheses. 35: 22; Collected Volumes, vol. 4, p. 148.
- ^ Bamberger, E. (1894). "Ueber die Reduction der Nitroverbindungen". Chemische Berichte. 27 (2): 1347–1350. doi:10.1002/cber.18940270229.
- ^ Bamberger, E. (1894). "Ueber das Phenylhydroxylamin". Chemische Berichte. 27 (2): 1548–1557. doi:10.1002/cber.18940270276.
- ^ Oxley, P. W.; Adger, B. M.; Sasse, M. J.; Forth1, M. A. (1989). "N-Acetyl-N-Phenylhydroxylamine Via Catalytic Transfer Hydrogenation of Nitrobenzene Using Hydrazine and Rhodium on Carbon". Organic Syntheses. 67: 187. doi:10.15227/orgsyn.067.0187.{{cite journal}}: CS1 maint: numeric names: authors list (link)
- ^ Kamm, O. (1925). "β-Phenylhydroxylamine". Organic Syntheses. 4: 57; Collected Volumes, vol. 1, p. 445. (download PDF)
- ^ Sone, T.; Hamamoto, K.; Seiji, Y.; Shinkai, S.; Manabe, O. (1981). "Kinetics and Mechanisms of the Bamberger Rearrangement. Part 4. Rearrangement of Sterically Hindered Phenylhydroxylamines to 4-Aminophenols in Aqueous Sulphuric Acid Solution". Journal of the Chemical Society, Perkin Transactions 2. 1981 (2): 1596–1598. doi:10.1039/P29810000298.
- ^ Kohnstam, G.; Petch, W. A.; Williams, D. L. H. (1984). "Kinetic Substituent and Isotope Effects in the Acid-Catalysed Rearrangement of N-Phenylhydroxylamines. Are Nitrenium Ions Involved?". Journal of the Chemical Society, Perkin Transactions 2. 1984 (3): 423–427. doi:10.1039/P29840000423.